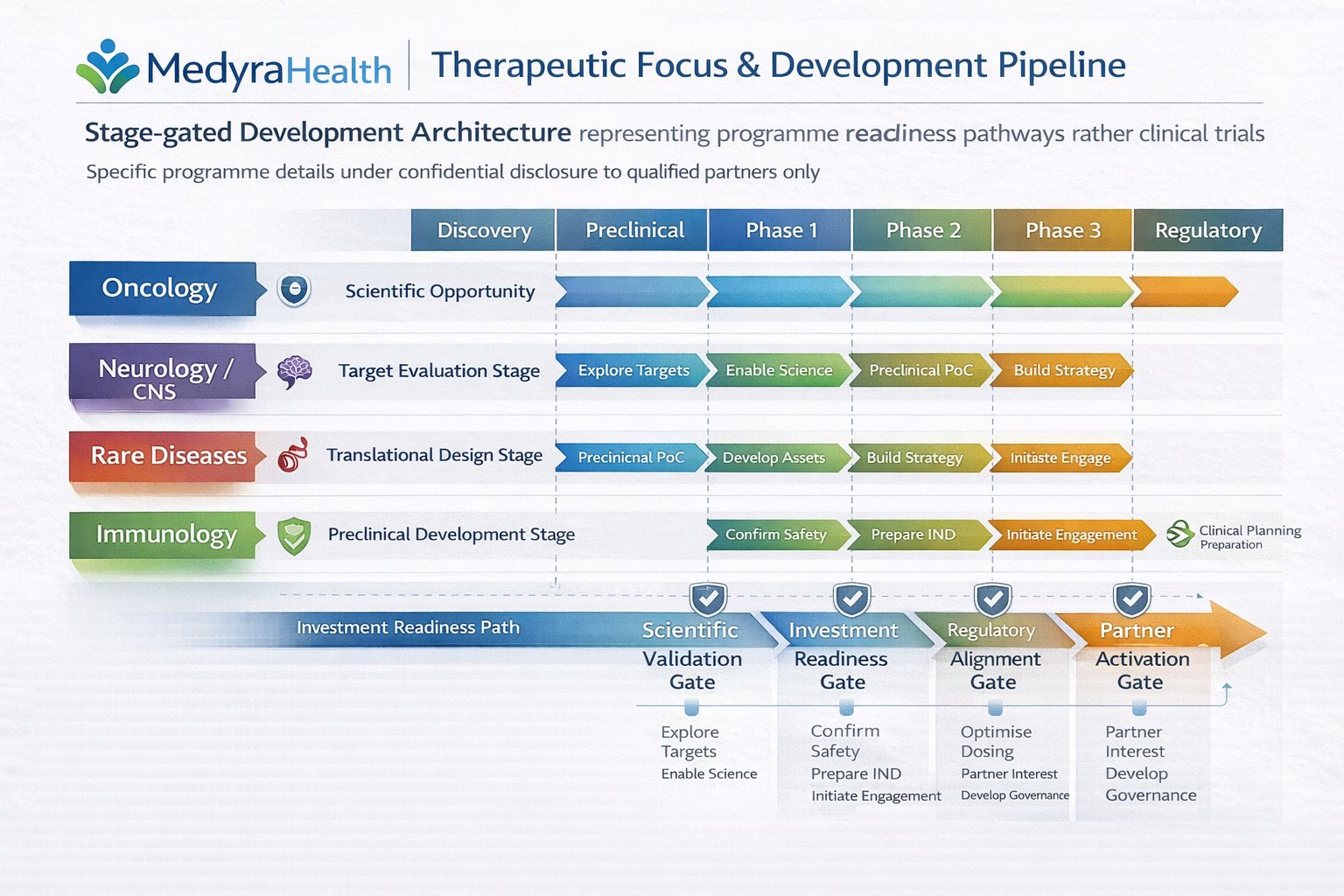
Pipeline overview
A high-level view of how Medyra Health can structure workstreams—from concept, through validation, to scalable delivery—without over-claiming.
Early stage
Target selection, data strategy, feasibility, and governance frameworks that enable controlled experimentation.
Mid stage
Clinical/operational intelligence, partner engagement, and evidence generation pathways aligned to real deployment.
Late stage
Scalable delivery planning: quality discipline, supply pathways, and measurable outcomes reporting.
Commercial readiness
Practical commercial packaging: positioning, portfolio clarity, and stakeholder-aligned reporting.
Early stage
Target selection, data strategy, feasibility, and governance frameworks that enable controlled experimentation.
Feasibility
Early-stage feasibility assessment is the most critical investment a development organisation can make. Medyra Health's feasibility frameworks address scientific feasibility (is the mechanism credible?), technical feasibility (can it be manufactured?), regulatory feasibility (is there a viable pathway?), and commercial feasibility (is there a viable business case?). All four dimensions must pass before a programme advances to mid-stage development.
Risk controls
Controlled experimentation at early stage requires robust risk identification, quantification, and mitigation frameworks. Medyra Health structures risk controls at a programme level—with a dedicated risk register, defined risk owners, and clear escalation pathways that ensure early-stage uncertainty is managed rather than ignored.
Roadmapping
Target selection, data strategy, and governance frameworks are aligned to a structured development roadmap that sets clear milestones, defines resource requirements, and establishes the criteria against which programme progression will be assessed. This roadmap is a living document—updated at each stage gate to reflect emerging scientific and operational insights.
images/pipeline-early.jpg
Mid stage
Clinical/operational intelligence, partner engagement, and evidence generation pathways aligned to real deployment.
images/pipeline-mid.jpg
Evidence
Mid-stage is where the evidence base for a programme is constructed. Medyra Health designs evidence generation pathways that are aligned to the specific regulatory, payer, and clinical requirements of the target indication—ensuring that every study conducted contributes to the development narrative in a structured, coherent way. We do not run studies speculatively; every piece of evidence is planned against a defined purpose and a clear audience.
Partnering
Clinical/operational intelligence and partner engagement are the defining activities of mid-stage development. Medyra Health maintains active engagement with potential commercial partners throughout the mid-stage development arc—ensuring that partnership discussions are informed by the most current programme data and that commercial alignment is established well before late-stage investment requirements intensify.
Operational readiness
Evidence generation pathways must be aligned to real deployment requirements—not just regulatory approval in isolation. Medyra Health's operational readiness frameworks assess manufacturing scalability, supply chain resilience, market access strategy, and healthcare system integration requirements at the mid-stage level, ensuring that late-stage and commercial activities are not constrained by operational unreadiness.
Late stage
Scalable delivery planning: quality discipline, supply pathways, and measurable outcomes reporting.
Quality discipline
Late-stage development demands the highest level of quality discipline—from GMP manufacturing compliance and process validation through to clinical trial data integrity and regulatory submission dossier quality. Medyra Health's quality frameworks are designed to meet the requirements of MHRA, EMA, and FDA review—without the bureaucratic excess that can slow programmes unnecessarily at this critical juncture.
Supply pathways
Scalable delivery planning requires supply pathway design that can flex from clinical trial supply volumes to commercial launch quantities—and then to full market demand—without supply disruption. Medyra Health designs supply pathways with dual-source capability, inventory buffering strategies, and logistics partner qualification frameworks that protect supply security across the critical launch window and beyond.
KPIs
Measurable outcomes reporting—structured around clinically meaningful endpoints, commercially relevant KPIs, and regulatory-required safety data—is the foundation of late-stage programme management. Medyra Health's KPI framework design ensures that performance is measured against what matters most: patient outcomes, partner confidence, and regulatory readiness.
images/pipeline-late.jpg
Commercial readiness
Practical commercial packaging: positioning, portfolio clarity, and stakeholder-aligned reporting.
images/pipeline-commercial.jpg
Positioning
Commercial positioning—establishing what a product is, who it is for, and why it is the right choice for that patient population—is the foundation of successful market entry. Medyra Health's commercial positioning frameworks are developed in close alignment with clinical evidence, payer requirements, and healthcare professional insights, ensuring that positioning is both clinically credible and commercially compelling.
Portfolio story
Portfolio clarity—presenting Medyra Health's development assets as a coherent, strategically aligned portfolio rather than a collection of disconnected programmes—is essential for investor confidence, partner engagement, and stakeholder trust. Medyra Health designs portfolio narratives that convey strategic intent, scientific coherence, and commercial ambition in a single, compelling story.
Stakeholders
Stakeholder-aligned reporting—presenting programme progress, commercial milestones, and risk status in formats tailored to each key stakeholder audience—is a core Medyra Health capability. We design reporting frameworks that give investors, partners, regulators, and clinical champions the information they need, in the format they expect, at the frequency they require.
Feasibility
Honest assessment before significant investment
Feasibility assessment is the most valuable and most under-invested phase of pharmaceutical development. Rigorous early feasibility — scientific, technical, regulatory, and commercial — dramatically reduces the probability of costly late-stage failure. Medyra Health conducts structured feasibility assessments across all four dimensions before committing any programme to full development investment.
Scientific feasibility
Biological target validation, mechanism of action credibility assessment, and competitive landscape analysis — establishing whether the scientific premise of a programme is sufficiently validated to justify development investment.
Technical & regulatory feasibility
Manufacturability assessment, formulation route feasibility, and regulatory pathway identification — confirming that a credible, cost-effective path from development concept to regulatory approval exists for each programme.
Risk controls
Managing uncertainty with structure and discipline
All pharmaceutical development involves risk. The question is not whether risk exists but whether it is identified, quantified, owned, and managed with appropriate rigour. Medyra Health's risk control frameworks establish a structured risk management discipline from the earliest stages of programme development — ensuring that every known risk has an owner, a mitigation plan, and a defined escalation threshold.
Risk identification
Systematic risk identification across scientific, technical, regulatory, commercial, and operational domains — using structured brainstorming, expert panel review, and analogous programme analysis to ensure completeness.
Risk mitigation
Defined mitigation actions, fallback options, and contingency plans for each identified risk — with clear ownership, timelines, and success criteria that enable risk mitigation progress to be tracked and reported objectively.
Roadmapping
A living plan that drives decisions and resources
A development roadmap is a structured, time-based plan that defines the key milestones, activities, resource requirements, and decision points for a programme from its current stage through to commercial launch. At Medyra Health, roadmaps are living documents — updated at each stage gate to reflect the current state of scientific, operational, and commercial knowledge — rather than static plans produced once and consulted rarely.
Milestone definition
Precise, measurable milestone definitions that create unambiguous shared understanding of what constitutes programme progress — preventing the "milestone inflation" that commonly distorts development reporting.
Resource planning
Resource requirement forecasting across FTE, CMO capacity, CRO capacity, and capital expenditure — enabling proactive resource acquisition rather than reactive scrambles that delay programme timelines.
Scenario planning
Best case, base case, and worst case scenario modelling that prepares programme teams and investors for the range of plausible development outcomes — and defines the decision rules for each scenario in advance.
Evidence generation
Building the scientific case that regulators and payers require
Evidence generation at mid-stage is the process of producing the clinical, preclinical, and real-world data required to support regulatory approval, health technology assessment, and commercial partner confidence. Medyra Health designs evidence generation strategies that are purposeful — every study is designed to answer a specific question for a specific audience, and contributes to a coherent overall evidence narrative.
Clinical study design
Proof-of-concept, dose-finding, and pivotal study design — including adaptive design approaches, biomarker-enriched enrolment strategies, and endpoint selection aligned to regulatory and HTA requirements.
Regulatory submission strategy
Early regulatory engagement (Type B meetings, scientific advice), regulatory strategy design, and submission dossier planning — aligned to MHRA, EMA, and FDA requirements in target markets.
Partnering
The right partner, at the right time, on the right terms
Strategic partnering — with global pharma, mid-size biotech, healthcare systems, technology companies, or impact investors — is a critical enabler of pharmaceutical development success. Medyra Health maintains active partnering engagement throughout the mid-stage development arc, ensuring that partnership discussions are informed by current data and that commercial alignment is established before late-stage investment requirements intensify.
Partner identification
Systematic partner landscape mapping, strategic fit assessment, and prioritised outreach planning — identifying the partners most likely to create genuine shared value with Medyra Health's programmes and capabilities.
Due diligence support
Data room preparation, scientific and clinical Q&A management, and regulatory document compilation — ensuring that partner due diligence processes run smoothly and create a positive impression of Medyra Health's programme quality and organisational capability.
Deal structuring
Term sheet development, deal structure optimisation, and commercial negotiation support — ensuring that partnership terms reflect the true value of Medyra Health's programmes while remaining commercially attractive to partner organisations.
Operational readiness
Prepared for the transition to late-stage and commercial operations
Operational readiness — the state of being genuinely prepared to execute late-stage development and commercial launch operations — is a function of decisions made years in advance. Medyra Health's operational readiness frameworks identify and address the manufacturing, supply chain, quality, and commercial infrastructure gaps that, if left unaddressed at mid-stage, become programme-delaying crises at late-stage.
Manufacturing readiness
CMO qualification status, process validation readiness, technology transfer planning, and capacity reservation frameworks — ensuring that manufacturing infrastructure is ready to support late-stage clinical supply and commercial launch.
Commercial infrastructure
Market access strategy development, health economic model construction, payer engagement planning, and launch team structuring — building the commercial foundation required to translate regulatory approval into rapid market uptake.
Quality discipline
GMP excellence from development through to commercial manufacture
Quality discipline in late-stage pharmaceutical development means operating to GMP standards across manufacturing, quality control, and clinical trial conduct — with the documentation rigour, change control discipline, and deviation management processes that regulatory authorities expect to see in a pre-approval inspection. Medyra Health's quality frameworks are designed to meet and exceed regulatory expectations across MHRA, EMA, and FDA jurisdictions.
GMP compliance
GMP compliance framework design, gap assessment against current regulatory expectations, and remediation programme management — preparing manufacturing operations for regulatory inspection at clinical and commercial scale.
QMS design
Quality management system design and implementation aligned to ICH Q10 — covering document control, training management, deviation handling, CAPA, change control, and quality risk management.
Regulatory inspection readiness
Pre-inspection readiness assessment, mock inspection programme management, and inspection response strategy — ensuring that regulatory inspections proceed smoothly and result in positive assessments.
Supply pathways
Uninterrupted supply from clinical trial to commercial launch and beyond
Supply chain failure at commercial launch — the moment when patient, prescriber, and investor expectations are at their highest — is a reputational and commercial catastrophe that no pharmaceutical organisation can afford. Medyra Health's supply pathway design frameworks are built to prevent this outcome, through multi-source API strategy, finished product inventory buffer planning, logistics partner qualification, and supply chain resilience testing that stress-tests the supply system before it is needed at scale.
Multi-source strategy
Dual or multi-source API and finished product manufacturing qualification — providing supply chain redundancy that protects against single-source failure, regulatory action, or force majeure events at critical supply nodes.
Launch supply planning
Pre-launch inventory build strategy, territory-by-territory launch supply forecasting, and distribution network design — ensuring that product is available in the right place at the right time from day one of commercial launch.
KPIs
Measuring what matters throughout development and beyond
Key performance indicators for late-stage pharmaceutical programmes must balance scientific rigour, operational efficiency, regulatory compliance, and commercial performance — providing a comprehensive and coherent picture of programme health to all stakeholder audiences. Medyra Health's KPI frameworks are designed to be meaningful, measurable, and manageable — capturing the metrics that genuinely predict programme success rather than the metrics that are merely easy to measure.
Clinical trial KPIs
Enrolment rate, data quality, protocol deviation rate, site performance, and patient retention metrics — enabling proactive trial management and early identification of execution risks before they impact programme timelines.
Manufacturing KPIs
Batch success rate, release cycle time, right-first-time rate, and stability performance metrics — providing real-time visibility of manufacturing quality and throughput performance at clinical and commercial scale.
Commercial KPIs
Market penetration rate, payer coverage, prescription volume, patient persistence, and net revenue metrics — tracking commercial launch performance against plan and enabling rapid commercial strategy adaptation where needed.
Positioning
Defining what you stand for in the market
Commercial positioning is the deliberate choice of what a product means to its target audiences — patients, prescribers, payers, and partners — and how it is differentiated from competing options. Effective positioning is grounded in clinical evidence, shaped by patient and prescriber insight, and tested for credibility with payer and HTA audiences before it is finalised. Medyra Health develops positioning frameworks that are both clinically credible and commercially compelling.
Differentiation strategy
Systematic competitive differentiation analysis that identifies the clinical, quality-of-life, convenience, and economic attributes on which a product can credibly claim superiority — and builds the evidence strategy required to substantiate those claims.
Message architecture
Core message development, claim hierarchy design, and supporting evidence mapping — creating a coherent, evidence-backed communication framework that can be adapted for prescriber, patient, payer, and partner audiences.
Portfolio story
A coherent narrative that creates more value than the sum of its parts
A portfolio story is the strategic narrative that presents a collection of development assets as a coherent, strategically aligned portfolio — demonstrating to investors, partners, and the broader market that Medyra Health's programmes are the product of a clear, differentiated scientific and commercial strategy, not a random collection of opportunistic bets. A compelling portfolio story creates a valuation premium over the sum of individual asset values.
Strategic narrative
Portfolio thesis development — articulating the unifying scientific, clinical, and commercial logic that connects Medyra Health's programmes and capabilities into a differentiated, investor-grade strategic narrative.
Investor communications
Investor deck design, pipeline visualisation, and financial narrative development — presenting the portfolio in a format and language that creates confidence and clarity for the full spectrum of pharmaceutical and healthcare investment audiences.
Stakeholders
Tailored engagement for every audience
Pharmaceutical development programmes involve a complex web of stakeholders — regulators, clinical investigators, patients, payers, commercial partners, investors, and the media — each with distinct information needs, engagement preferences, and influence over programme outcomes. Medyra Health's stakeholder engagement frameworks ensure that the right information reaches the right audience in the right format at the right time — building the trust, confidence, and advocacy that successful programmes require.
Regulatory engagement
Pre-submission meeting strategy, scientific advice request design, and regulatory authority relationship management — building the constructive, transparent relationships with MHRA, EMA, and FDA that accelerate review timelines and reduce approval uncertainty.
Clinical champion development
Key opinion leader identification, scientific advisory board design, and clinical investigator network development — building the clinical advocacy base required for successful trial execution and post-approval prescriber adoption.
Investor relations
Investor presentation design, pipeline milestone communication, and financial narrative management — maintaining investor confidence through the inevitable uncertainties and setbacks of pharmaceutical development.